 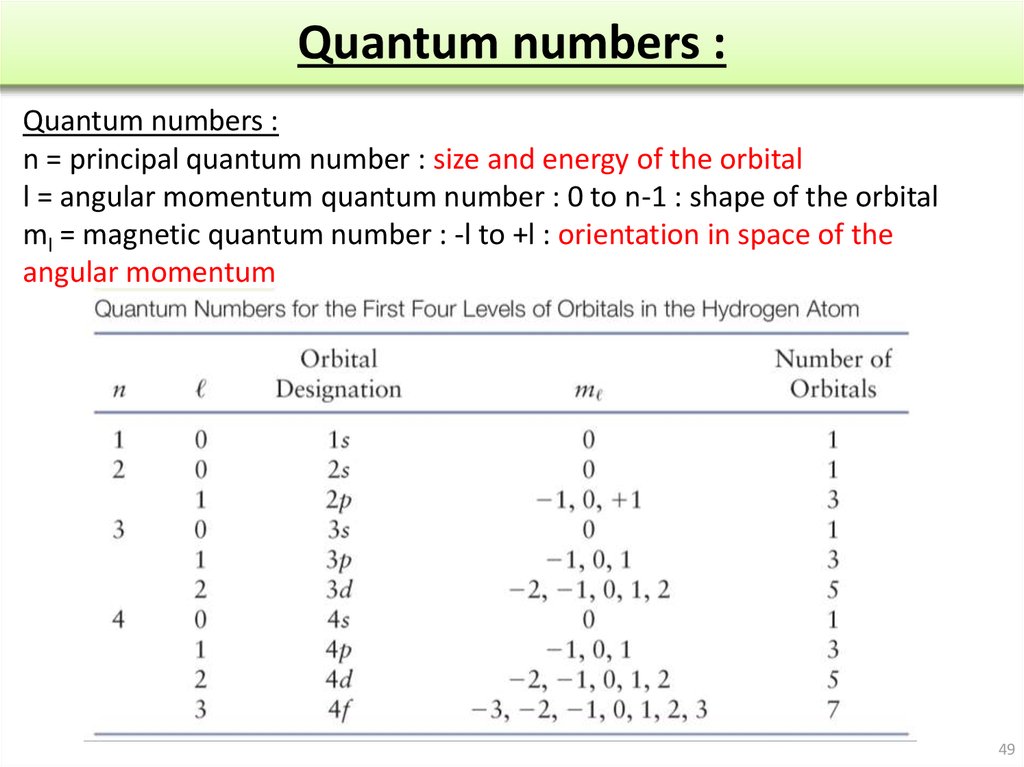
If your institution is not listed or you cannot sign in to your institution’s website, please contact your librarian or administrator.Įnter your library card number to sign in. Following successful sign in, you will be returned to Oxford Academic.Do not use an Oxford Academic personal account. When on the institution site, please use the credentials provided by your institution.Select your institution from the list provided, which will take you to your institution's website to sign in.Click Sign in through your institution.Shibboleth / Open Athens technology is used to provide single sign-on between your institution’s website and Oxford Academic. This authentication occurs automatically, and it is not possible to sign out of an IP authenticated account.Ĭhoose this option to get remote access when outside your institution. Typically, access is provided across an institutional network to a range of IP addresses. If you are a member of an institution with an active account, you may be able to access content in one of the following ways: Get help with access Institutional accessĪccess to content on Oxford Academic is often provided through institutional subscriptions and purchases. It was only following the introduction of the Pauli exclusion principle and the development of the new quantum mechanics that Heisenberg succeeded where everyone else had failed. even the astronomers have not yet satisfactorily solved the three-body problem in spite of efforts over the centuries.†A succession of the best minds in physics, including Hendrik Kramers, Werner Heisenberg, and Arnold Sommerfeld, made strenuous attempts to calculate the spectrum of helium but to no avail. As one physicist stated, “We should not be surprised. But when it came to trying to predict quantitative aspects of atoms, such as the ground-state energy of the helium atom, the old quantum theory was quite hopeless. An explanation could be given for the periodic table in terms of numbers of electrons in the outer shells of atoms, but generally only after the fact. The old quantum theory was quantitatively impotent in the context of the periodic table since it was not possible to even set up the necessary equations to begin to obtain solutions for the atoms with more than one electron. 
It was essentially a chemical fact that was accommodated in terms of the quantum mechanical understanding of the periodic table. Moreover, despite the rhetoric in favor of quantum mechanics that was propagated by Niels Bohr and others, the discovery that hafnium was a transition metal and not a rare earth was not made deductively from the quantum theory. Main Smith, and Charles Bury were able to go further than physicists in assigning electronic configurations, as described in chapter 8, because they were more familiar with the chemical properties of individual elements. Indeed, in several cases, chemists such as Irving Langmuir, J.D. Although the development of this theory provided a way of reexpressing the periodic table in terms of the number of outer-shell electrons, it did not yield anything essentially new to the understanding of chemistry. In chapter 7, the influence of the old quantum theory on the periodic system was considered. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
